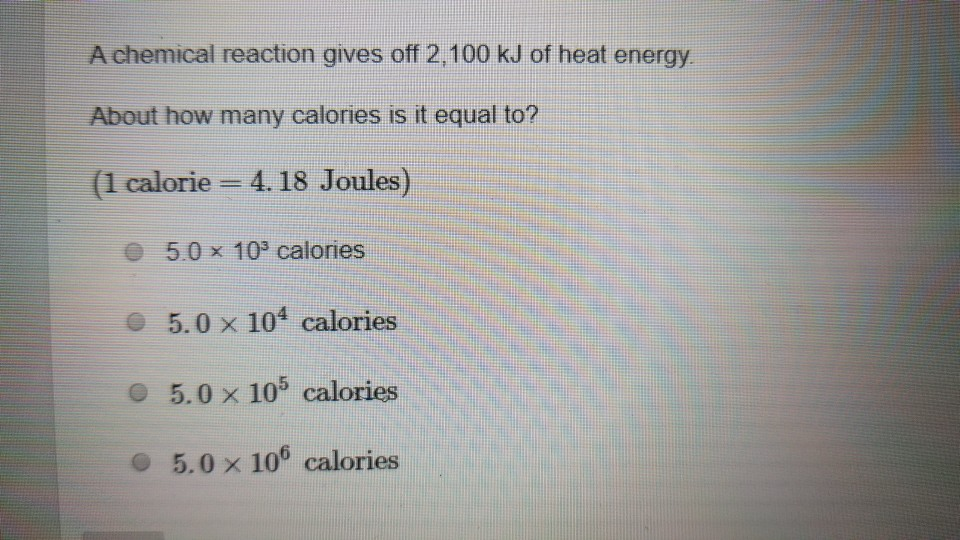Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download

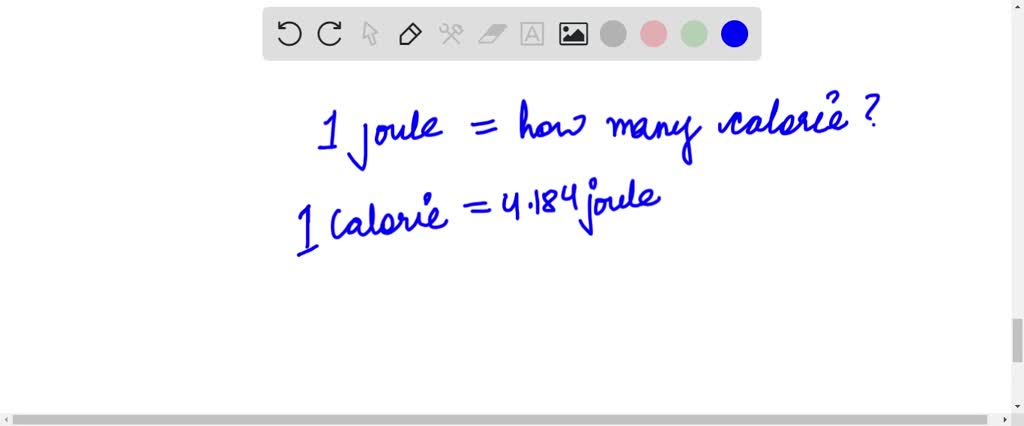
A calorie is a unit of heat or energy and it equals about 4.2 J where 1J = 1 kg m2s–2. Suppose we - Brainly.in

The unit of Mass is alpha kg. The unit of length is beta metre and the unit of time is gamma second. The magnitude of calorie in the new system is:[1 calorie =
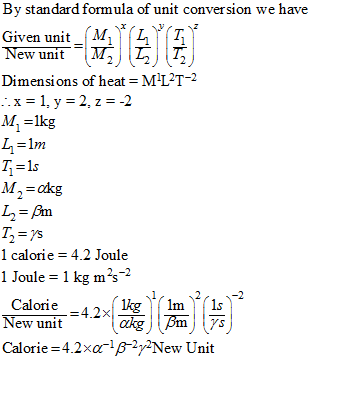
A calorie is a unit of heat or energy and it equals about 4 2 J where 1J = 1 - Physics - Units And Measurements - 2701112 | Meritnation.com

Thermochemistry! AP Chapter 5. Temperature vs. Heat Temperature is the average kinetic energy of the particles in a substance. Heat is the energy that. - ppt download

One calorie of heat is equivalent to 4.2 J . One BTU (British thermal unit) is equivalent to 1055 J . The value of one BTU in calorie is?
A calorie is a unit of heat or energy and it equals about 4.2 J where 1J = 1 kg m^2s^–2. - Sarthaks eConnect | Largest Online Education Community
![SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ] SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ]](https://cdn.numerade.com/ask_images/f3f65c4b70424578aa674a37eabc4800.jpg)
SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ]

Not all calories are equal – a dietitian explains the different ways the kinds of foods you eat matter to your body

Goals of Chapter Assess heat transfer associated with changes in temperature and changes of state. Apply the First Law of Thermodynamics. Define and understand. - ppt download