Isotopes. Isotopes of Magnesium Atomic symbol Mg Mg Mg Number of protons Number of electrons Mass number Number of neutrons ppt download
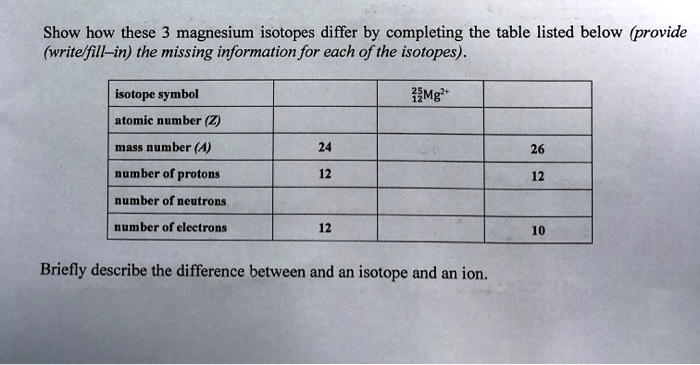
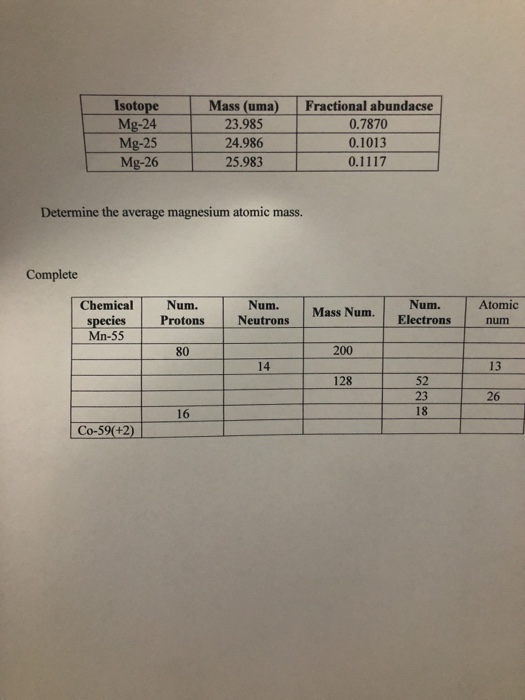
SOLVED: Show how these 3 magnesium isotopes differ by completing the table listed below (provide (writelfill-in) the missing information for each of the isotopes) . isotope symbol izMg + atomic number (2)
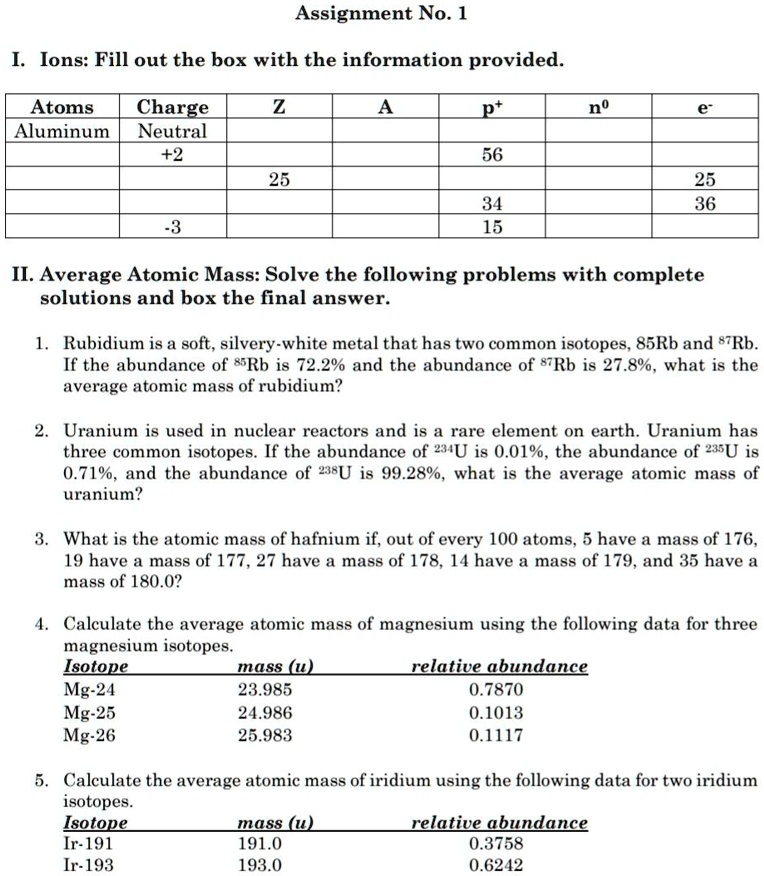
SOLVED: Assignment No. 1 Ions: Fill out the box with the information provided: Atoms Charge Aluminum Neutral +2 56 25 25 36 34 15 II Average Atomic Mass: Solve the following problems

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube

Do Now 1.What is the atomic number of copper (Cu) 2.What element has 80 protons 3.An isotope of magnesium (Mg) has a mass number of 26. How many neutrons. - ppt download

Do Now 1.What is the atomic number of copper (Cu) 2.What element has 80 protons 3.An isotope of magnesium (Mg) has a mass number of 26. How many neutrons. - ppt download









:max_bytes(150000):strip_icc()/GettyImages-1135707671-640473b29d534e15a24491c0d6b2789e.jpg)