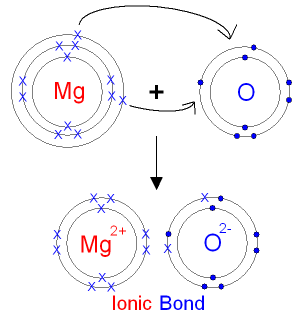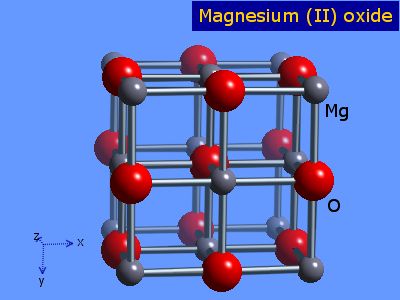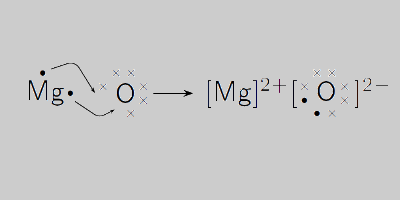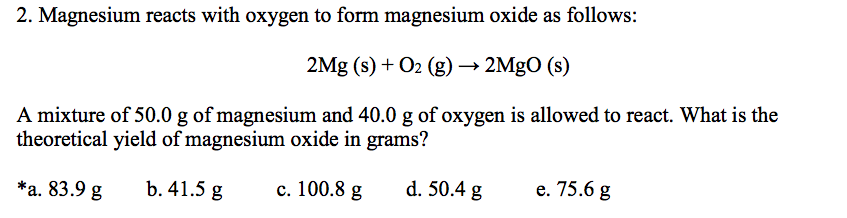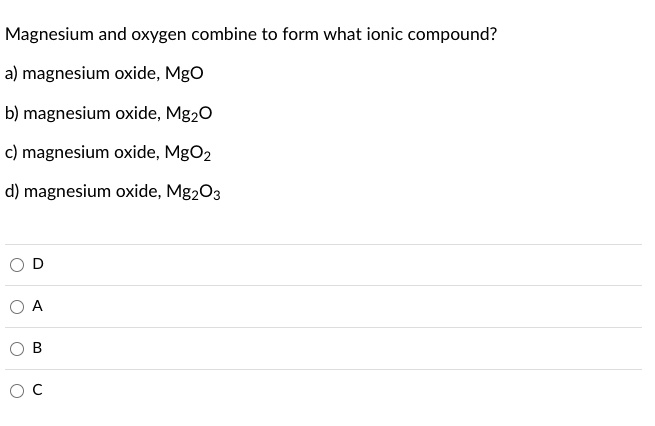
SOLVED: Magnesium and oxygen combine to form what ionic compound? a) magnesium oxide, Mgo b) magnesium oxide, MgzO magnesium oxide; MgOz magnesium oxide; MgzO3

Diagram to show ionic bonding in magnesium oxide, Stock Vector, Vector And Low Budget Royalty Free Image. Pic. ESY-057091086 | agefotostock

Magnesium and oxygen combine in the ratio of `3 : 2` by mass to form magnesium oxide. What ma - YouTube
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as

Question Video: Calculating the Mass of Oxygen Required to React with a Given Number of Magnesium Atoms | Nagwa
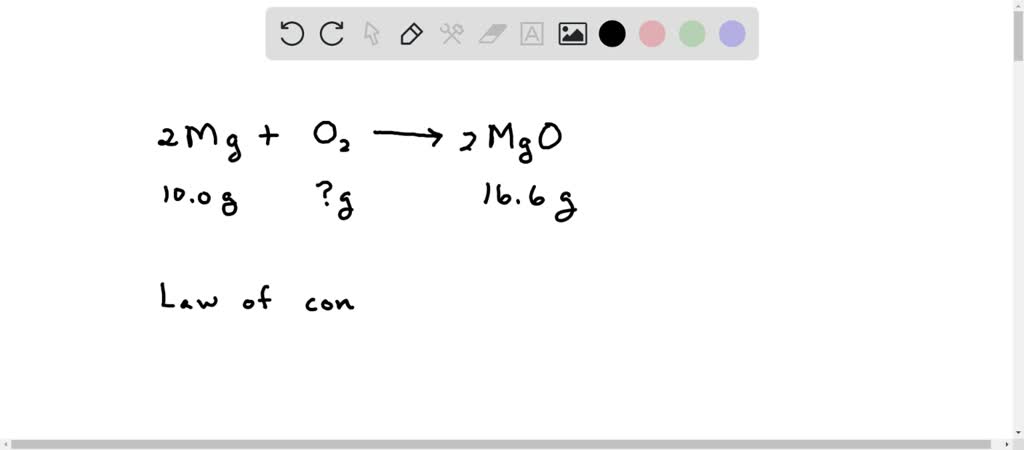
SOLVED: A 10.0-g sample of magnesium reacts with oxygen to form 16.6 g of magnesium oxide. How many grams of oxygen reacted?